A COVID-19 vaccine developed by a Chinese company has proven to be effective on a species of monkey during trial testing. Researchers from Sinovac Biotech conducted the trial on rhesus macaques back in April. The study was published on April 19, and a peer-reviewed version was recently released on May 6.
The research team gave two separate doses of the vaccine, which is a chemically inactivated version of the virus, to eight rhesus macaques. After three weeks, the group was introduced to SARS-CoV-2, which causes the COVID-19 disease, and found that none developed a serious infection. The study showed that monkeys given the highest dose of vaccine responded the best, showing no signs of COVID-19 in the pharynx or lungs, while those with lighter doses had a “viral blip,” according to Science magazine. Meanwhile, unvaccinated monkeys given the virus showed clear-cut COVID-19-like symptoms.
Human trials for the vaccine began on April 16.
Given the method used to develop the vaccine, virologist Florian Krammer of the Icahn School of Medicine at Mount Sinai, said, “many vaccine producers, also in lower-middle-income countries, could make such a vaccine.”
However, there are still reservations about the trial results, such as the small sample size and whether there is a better animal model for studying the coronavirus.
Beijing-based Sinovac started phase I clinical trials in Jiangsu province, and phase II studies may begin as soon as mid-May. Of the more than 100 current COVID-19 vaccine projects, eight are being tested on people – including four in China, according to media outlet Stat.
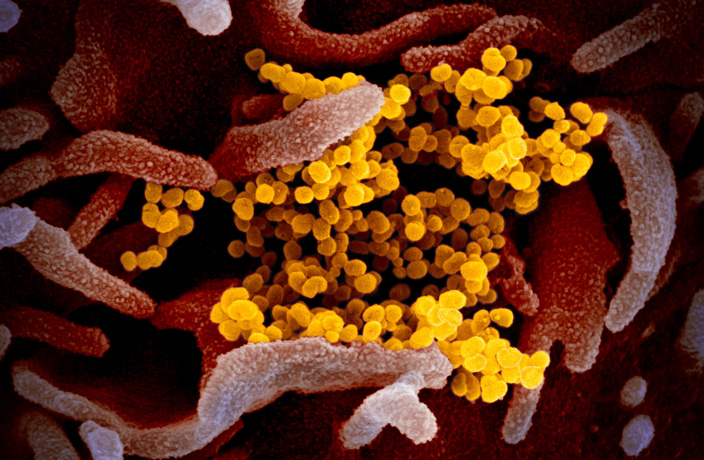
[Cover image via NIAID-RML]




















0 User Comments